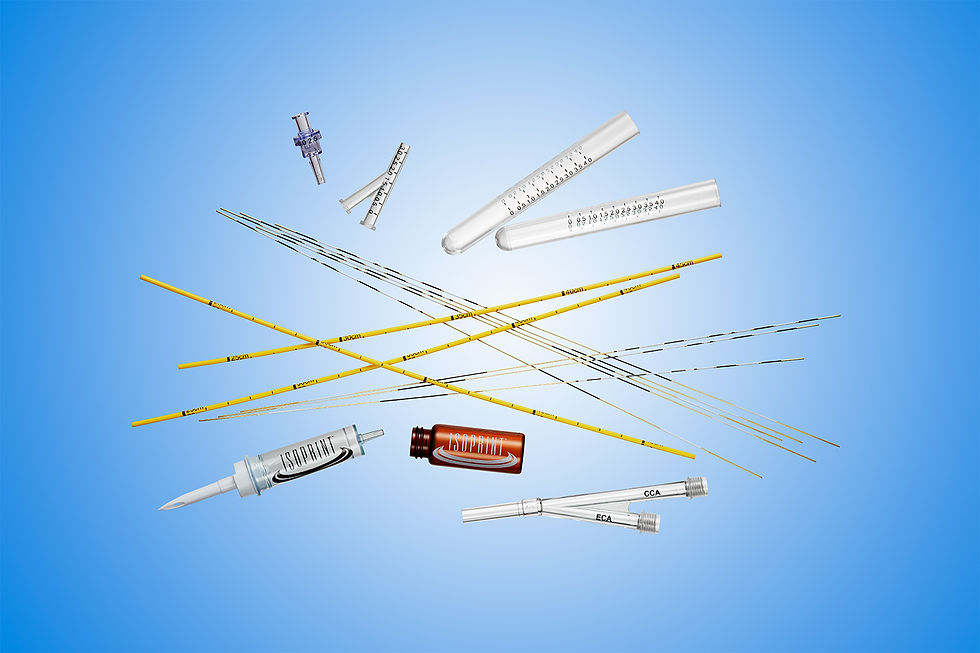
Isoprint Medical Pad Printing Capabilities
Isoprint specializes in medical component pad printing for original equipment manufacturers (OEM). We use Class VI medical grade inks to meet biosafety concerns and use substrate-specific surface treatment for permanent ink adhesion. We have environmentally controlled systems for clean manufacturing (Class 7 cleanroom) and we are ISO 9001:2015 certified.
Our clientele includes small independent firms developing prototypes to production runs for Fortune 500 companies.
Our capabilities include:
-
Highly detailed and accurate image reproductions
-
Permanent imprint adhesion that withstands various sterilization techniques and will not flake or rub off
-
Pad printing on all substrates and on atypical surfaces and unusual device shapes and sizes
-
Pad printing using radio opaque and conductive inks
-
360° surface imprints
-
Custom color and multi-color requirements
-
Use of Class VI medical grade inks to meet biosafety concerns
-
Use of substrate-specific surface treatment for permanent ink adhesion
-
Environmentally controlled systems for clean manufacturing
-
Leveraging the latest tooling designs for production consistency and economical costing.


A sampling of medical products we can pad print on:
-
Catheters and microcatheters (i.e., colonoscopy, dialysis, EVD, intrauterine, intravascular, neonatal umbilical, thoracic, trocar, ventriculostomy)
-
Clips and adapters
-
Connectors
-
Guide wires
-
Lenses
-
Shafts
-
Syringes
-
Tubing (i.e., endotracheal, feeding, hypo, jejunostomy, tracheal)
-
Products with unusual or atypical surfaces
Substrates we print on
Using Class VI medical grade inks and process specific pre-treatment techniques, we are able to achieve excellent ink adhesion and print quality on a wide variety of substrates. Our pad printing will withstand various sterilization techniques.
Acetal Copolymer
Acrylic (PMMA)
Acrylonitrile Butadiene Styrene (ABS)
Aluminum
Carbothane
Cotton and other textiles
Delrin
Dow Isoplast 2531
Dupont Delrin
Dupont Elvax 3165
Elastollan (thermoplastic polyurethane)
Estane Expanded Polytetrafluoroethylene (ePTFE)
Flourinated ethylene-propylene (FEP)
Fabrics (surgical and implantable)
Glass
Grilamid (nylon)
Metals
Neoprene
Nitinol
Nylon
Paper
Pebax
Pellethane
Perfluoroalkoxy (PFA)
PET Shrink Tubing
Polyamide
Polycarbonate (PC)
Polyester
Polyethermide
Polyethylene (PE)
Polyetheretherketones (PEEK)
High Density Polyethylene (HDPE)
Low Density Polyethylene (LDPE)
Polyimide (PI)
Polyolefin
Polypropylene (PP)
Polystyrene (PS)
Polytetrafluoroethylene (PTFE)
Polyurethane
Polyvinylchloride (PVC)
Powder coated metals
Santoprene
Silicone
Stainless Steel
Styrene-Acrylonitrile Copolymer (SAN)
Tecoflex
Textiles (surgical and implantable)
Ultem (polyethermide)
Urethane
Wood

Medical Grade Inks
We have carefully selected the following biocompatible and medical grade inks for use in our printing processes. Our team can help you select the appropriate ink to use for your project and its application. Technical data sheets and material safety data sheets (MSDS) are available upon request.

Radio Opaque Ink
Conductive Ink
We use conductive ink for products requiring conductivity in their inks.
Radio Opaque Ink
We use printable radio opaque ink for products that require internal medical device markings that can be seen using medical imaging equipment.
Silicone Inks
Silicone inks are designed for printing on silicone rubber parts.
High-gloss, quick curing two-component ink
Features outstanding mechanical and chemical resistance and excellent adhesion on critical materials.

Plasma Gas Treatment
When required, we use plasma gas treatment to allow for permanent printing adherence to certain substrates such as silicone, polyurethane, Delrin®, fluorinated ethylene-propylene (FEP), polyethylene (PE), polypropylene (PP), and polytetrafluoroethylene (PTFE). Plasma gas treatment is a process that uses gas and electricity instead of heat as a pretreatment of a surface to be printed on.
